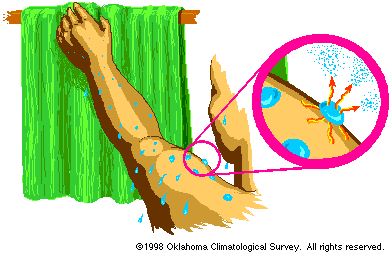|
|
Heat Transfer
|
|
|
|
|
|
|
Principle
|
The driving force for atmospheric motion
is the sun and, in particular, the uneven distribution of
solar radiation across the earth. It is the primary job of
the atmosphere to redistribute energy in order to achieve a
balance from pole to equator. We need to understand methods
of heat exchange in the atmosphere to appreciate how the
thin atmosphere keeps us alive.
|
|
|
|
|
|
|
Energy
|
|
|
|
|
|
|
|
Figure 1- Evaporation and Condensation
|
|
|
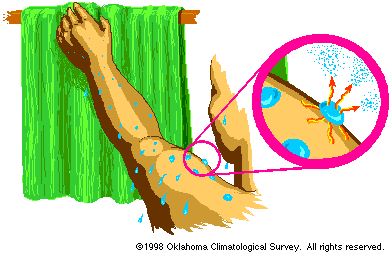
|
|
|
Latent Heat
|
- Latent heat is the heat energy required to change a
substance from one state to another.
There are basically three states
of matter: solid, liquid, and gas. The difference between
them is how the molecules are arranged. Solids have
tightly-packed molecules, liquids are still bound
together but not strongly enough to keep them from
flowing, and gas molecules are free-flowing, not bound to
one another at all. Energy is required to change from one
state to another because bonds must be loosened, broken,
tightened, or made. Energy must be given to the molecules
if bonds are to be loosened or broken and taken from the
molecules if they are to be tightened or made.
- Energy is required to change from solid to liquid,
liquid to gas (evaporation), or solid to gas
(sublimation). Energy will be released to change from
liquid to solid (fusion), gas to liquid (condensation),
or gas to solid.
- Latent heat of evaporation is the energy used to
change liquid to vapor.
IMPORTANT: The temperature does
not change during this process, so heat added goes
directly into changing the state of the substance. About
600 calories of energy are needed for every gram of water
at room temperature. This is why you cool when you step
out of the shower. Heat is taken from your skin to
evaporate the water on your body.
- Evaporation is a cooling process.
- Latent heat of condensation is energy released when
water vapor condenses to form liquid droplets.
An identical amount of calories
(about 600 cal/g) is released in this process as was
needed in the evaporation process. This is one mechanism
of how thunderstorms maintain their intensity. As moist
air is lifted and cooled, water vapor eventually
condenses, which then allows for huge amounts of latent
heat energy to be released, feeding the storm.
- Condensation is a warming process.
- Latent heat of fusion describes both changing from
solid to liquid and from liquid to solid.
From solid to liquid, about 80
calories per gram are needed. From liquid to solid, about
80 cal/g are released.
- Latent heat of sublimation describes both changing
from solid to gas and gas to solid.
Sublimation is rare as compared
to the other changes of state. From solid to gas 600 + 80
= 680 calories per gram are needed. From gas to solid,
680 cal/g are released.
|
|
|
|
|
|
|
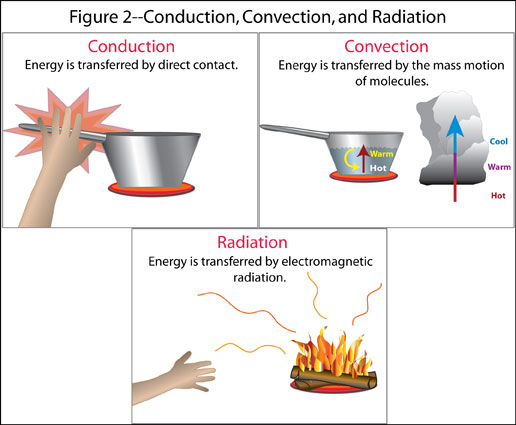
Figure 2- Conduction, Convection and Radiation
|
|
|
|
|
|
Methods of Heat Transfer
|
- Conduction- energy is transferred by the direct
contact of molecules, not by the movement of the material
Example: putting your hand on a stove burner. The
amount of energy transferred depends on how conductive
the material is. Metals are good conductors, so they are
used to transfer energy from the stove to the food in
pots and pans. Air is the best insulator, so good
insulating products try to trap air and not allow it to
move.
- Convection- energy is transferred by the mass motion
of groups of molecules resulting in transport and mixing
of properties
Example: holding your hand over a stove burner. In
meteorology, we speak of convection predominantly as that
caused by rising currents of warm air. We refer to all
other mass motions of air as advection.
- Radiation- energy is transferred by electromagnetic
radiation
Example: heat felt when standing away from a large
fire on a calm night. Everything that has a temperature
above absolute zero radiates energy. Radiation is not
"felt" until it is absorbed by a substance. It does not
require a medium to transfer energy through as do
conduction and convection.
|
|
|
|
|
|
|
Specific Heat
|
- Specific heat is the amount of heat needed to raise
the temperature of one gram of a substance one degree
Celsius.
- The specific heat of water is very high compared to
other substances, so water can store energy longer than
most other substances.
For example, the Gulf of Mexico
remains warm during the night, when air and soil
temperatures decrease rapidly.
Why is the Southern Hemisphere
summer generally not warmer than the Northern Hemisphere
summer although Earth is closer to the sun during the
Southern Hemisphere summer? Because most of the Southern
Hemisphere is water, which regulates the seasonal
temperatures.
|
|
|
|
|
|
|
End
|
|